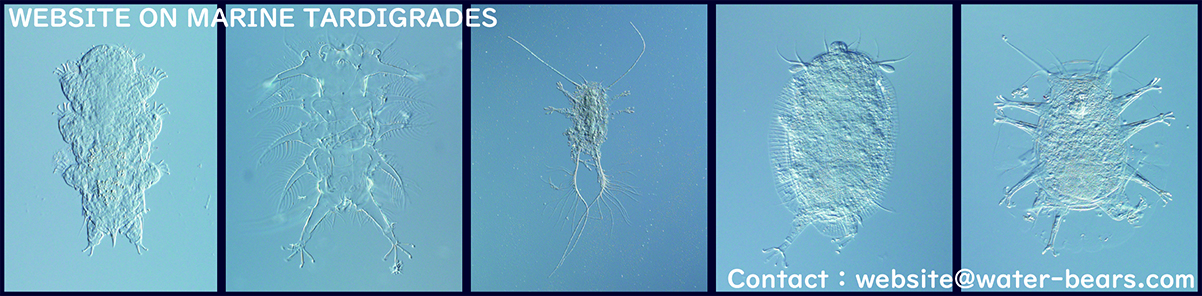
How to catch them
Beginners
Go to a rocky shore or a beach and scrape some barnacles off rocks (small barnacles are good). You never know where they are so take samples at different levels, e.g. near the water, mid-tide level, and near the high-water mark. When you are back at home/lab, place the barnacles in a dish with sea water and leave it for a while and observe the bottom of the dish and pieces of barnacles in the dish with a dissecting microscope. There you may find some crawling Echiniscoides, a marine tardigrade typically found from barnacles (see also Where are they?). They are probably one of the smallest organisms you see in the dish so take your time and observe closely at high magnification (at least 30x). If you want tou extract tardigrades more efficiently, see the next section.
Intermediate
Sand is the main habitat of marine tardigrades. See Where are they? for finding "good" sand. The standard method of extracting the marine tardigrades from the sediment (also barnacles and other substrates) is the osmotic shock/freshwater shock method. You could read how effective the method is in Kristensen (1983).
Advanced
If you try the freshwater shock method for mud or clay samples, you would notice that the fine particles never settle (actually they settle after 15 minutes or longer). This is a big problem for the method because you cannot separate animals and those particles, both floating in the water column and animals might as well settle at the bottom before the mud particles settle.
To avoid this problem there is the floatation method (desity separation method) using sugar in the old day and now colloidal silica (I use colloidal silica HS-40?). Instead of water stir the sediment with this reagent (to save reagent, you could diluted approximately 1:1 with tapwater, if you are not sure, just use the thick concentration of the reagent) and cetrifuge for a minute or so or keep it still for a while. The animals would be floating in the top layer of the supernatant separated from the sediment. This method uses the difference in density of animals and sediment. If the animals are dirty (covered with sediment particles) or have shells they may be with the sediment.
For this method, the sediment has to be washed with freshwater before being mixed with the reagent. The reagent becomes a gel with sea water (and also formaldehyde and ethanol).
This method could be used for sand too but some tardigrades are dirty so I recommend using the floatation method only with mud and clay. Further, it is expensive to process bulks of samples with this method.
ü@ü@

Coarse sand with/without tardigrades

Higgins' mermaid bra.

Feel the tardigrades in the dish?